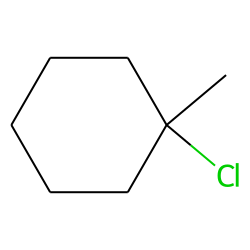
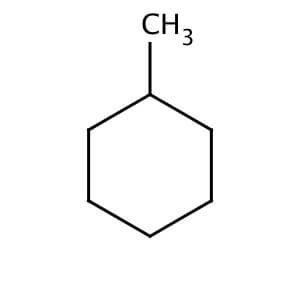
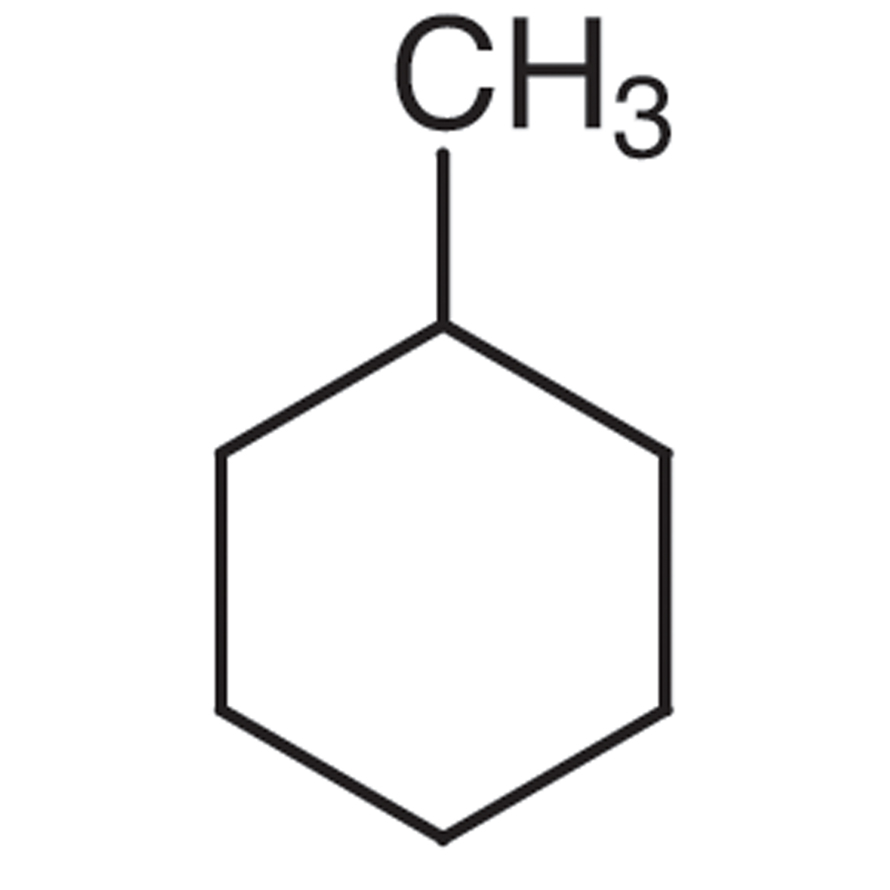
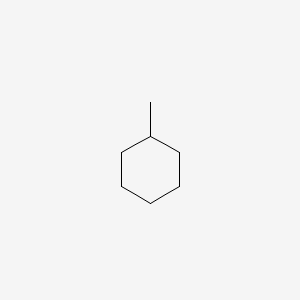
Name of the alkene which will yield/chloro 1-methylcyclohexane by its reaction with HCl. Write the - YouTube

Assume that you are carrying out the dehydration of 1-methylcyclohexanol to yield 1-methylcyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete? | Homework.Study.com
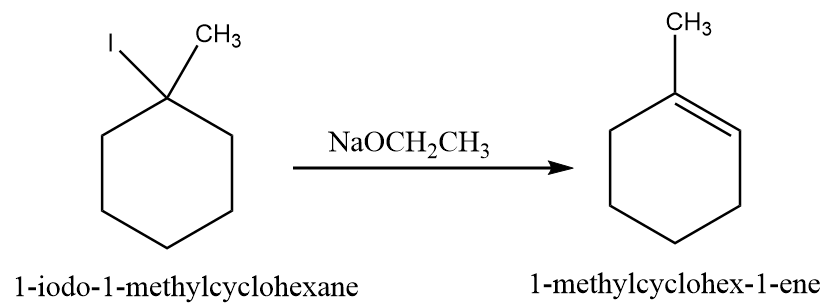
When 1-iodo-1-methylcyclohexane is treated with $ NaOC{H_2}C{H_3} $ as the base, the more highly substituted alkene product predominates. When $ KOC{(C{H_3})_3} $ is used as the base, then less substituted alkene predominates.

1-isopropenyl-1-methylcyclohexane Molecular Weight - C10H18 - Over 100 million chemical compounds | CCDDS
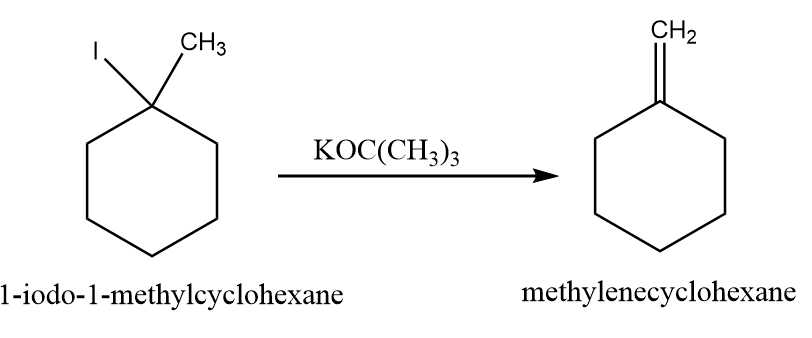
When 1-iodo-1-methylcyclohexane is treated with $ NaOC{H_2}C{H_3} $ as the base, the more highly substituted alkene product predominates. When $ KOC{(C{H_3})_3} $ is used as the base, then less substituted alkene predominates.

Write the structure of the major organic product formed in the reaction of 1 -methylcyclohexane with hydrogen chloride. | Homework.Study.com