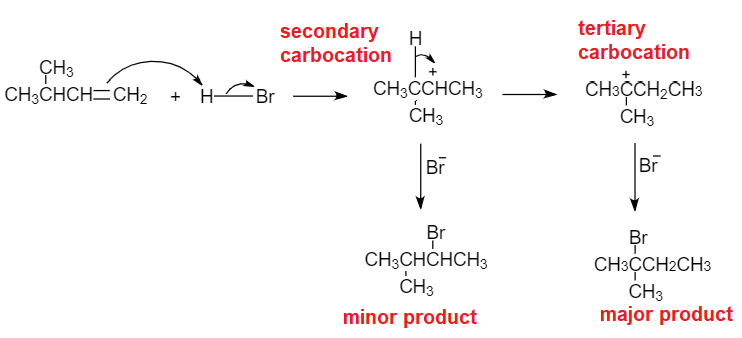
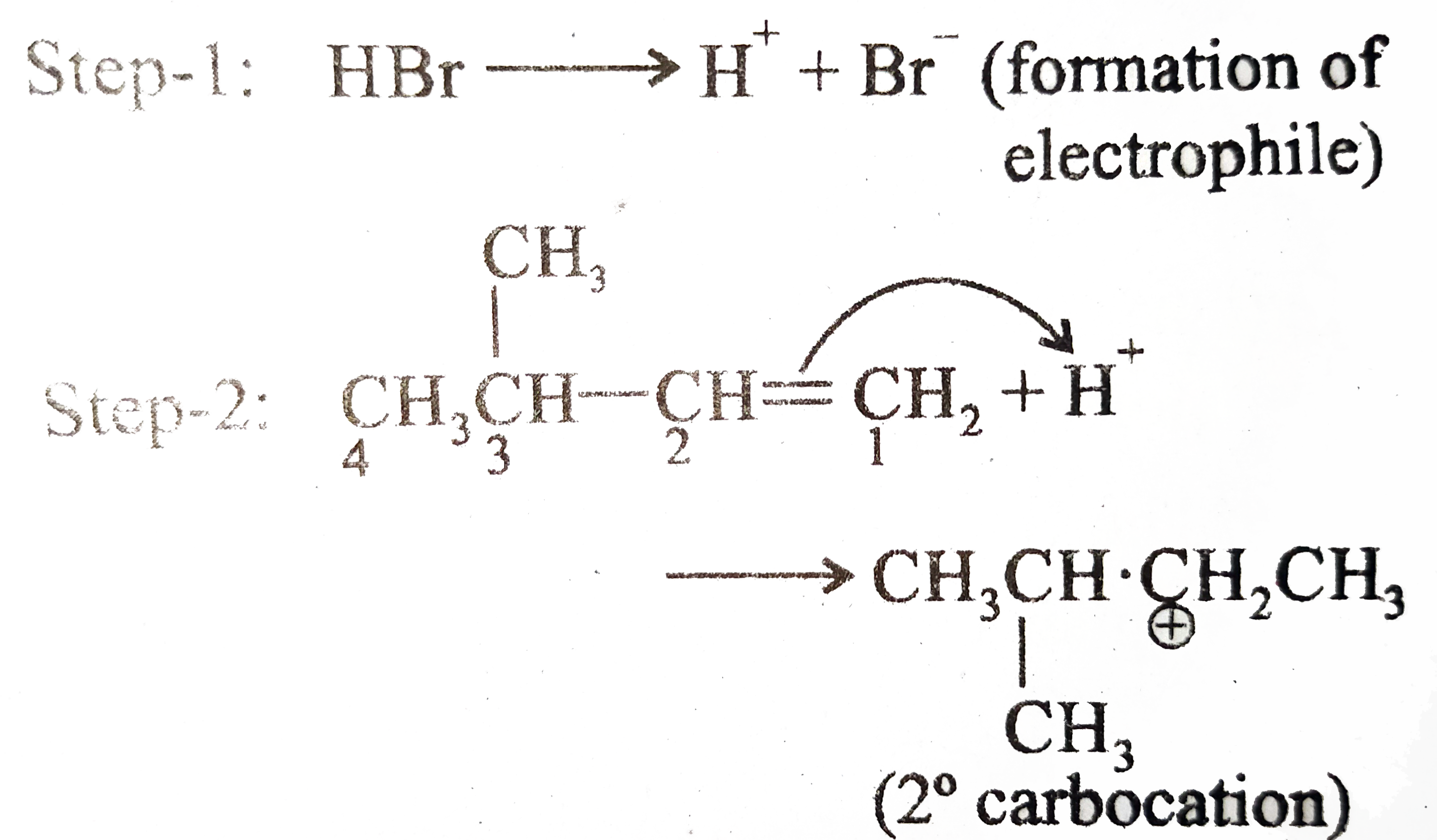
Predict the major and minor products of 2-methyl-2-butene with HBr as an electrophilic addition reaction. Include the intermediate reactions. | Homework.Study.com

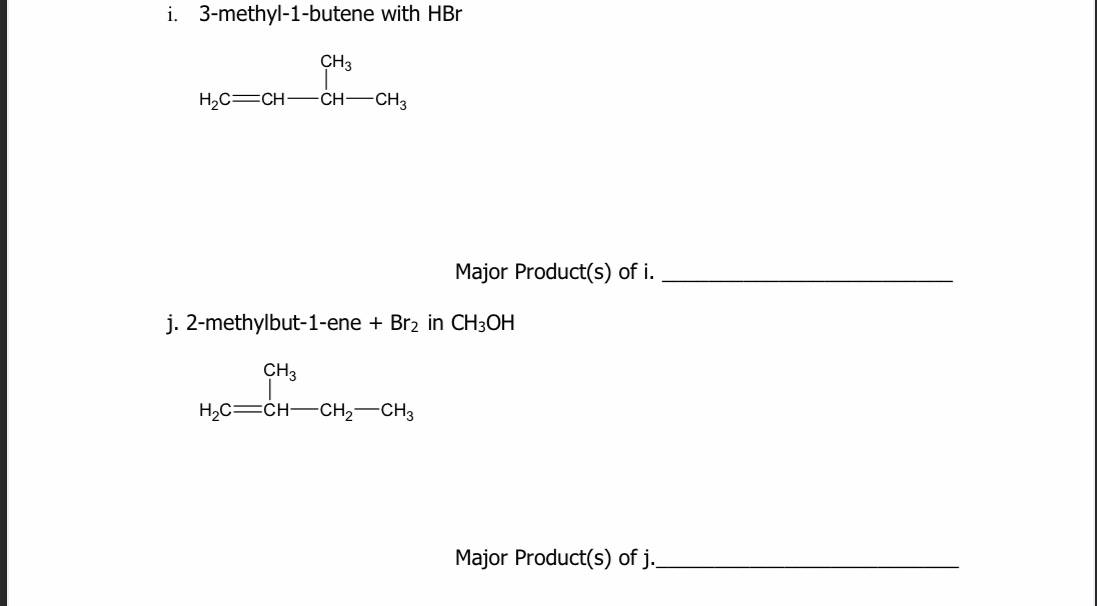
When 2-methyl-1-butene is treated with HBr, only one product forms: 2-bromo-2-methylbutane. Why? A. The reactants have read the published papers of Vladimir Markovnikov, and dutifully obey his rule. B. 2-bromo-2-methylbutane is the

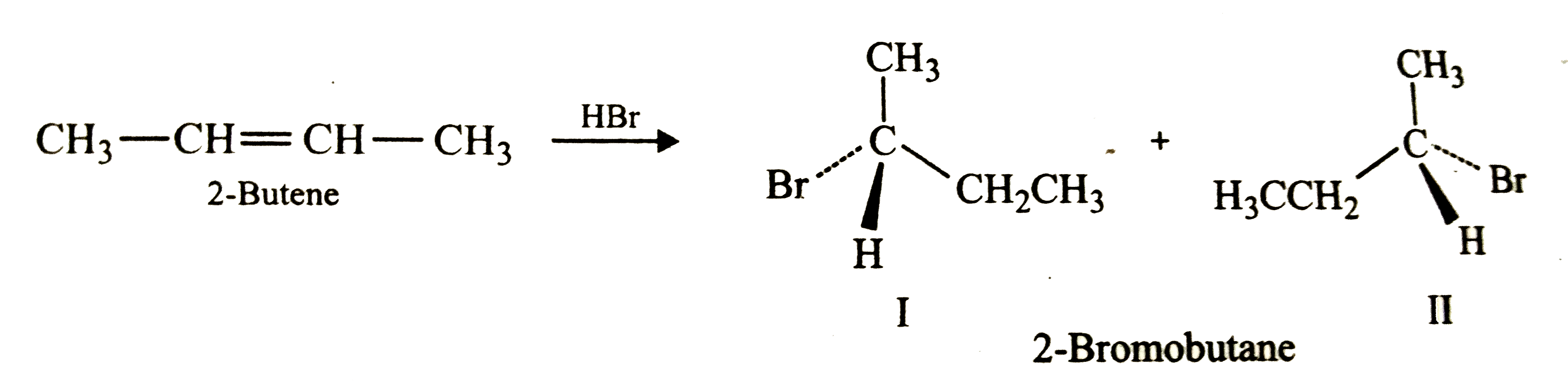
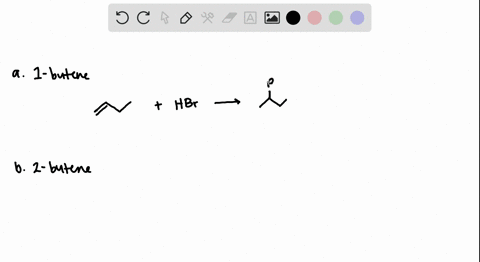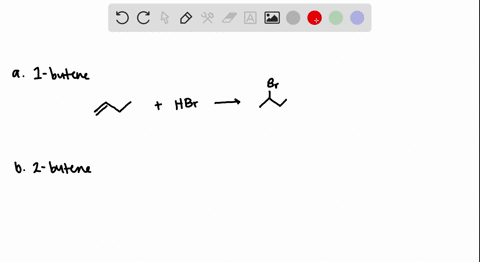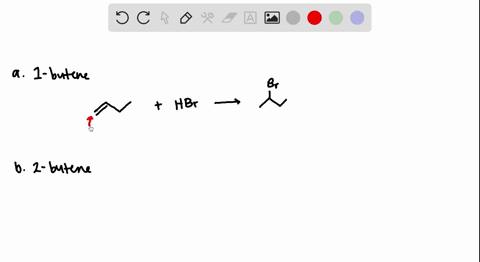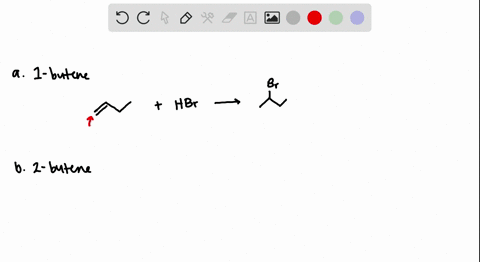
The addition of HBr to 1-butene gives a mixture of products (I),(II) and (III):The mixture consists of :

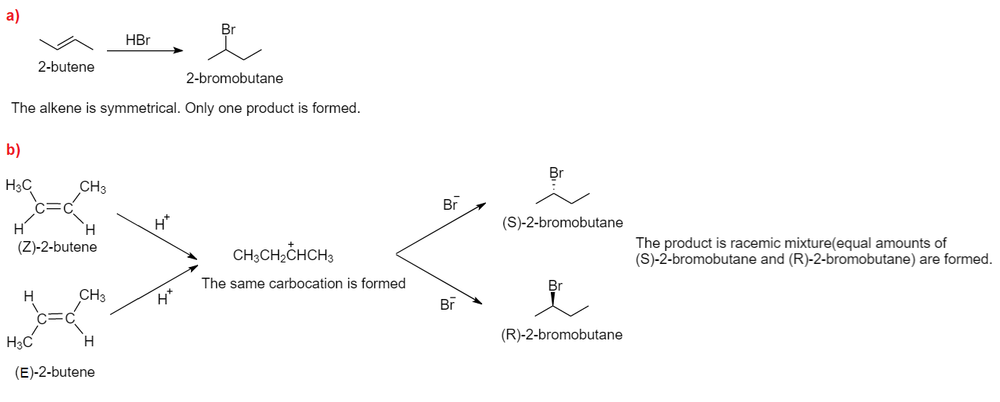
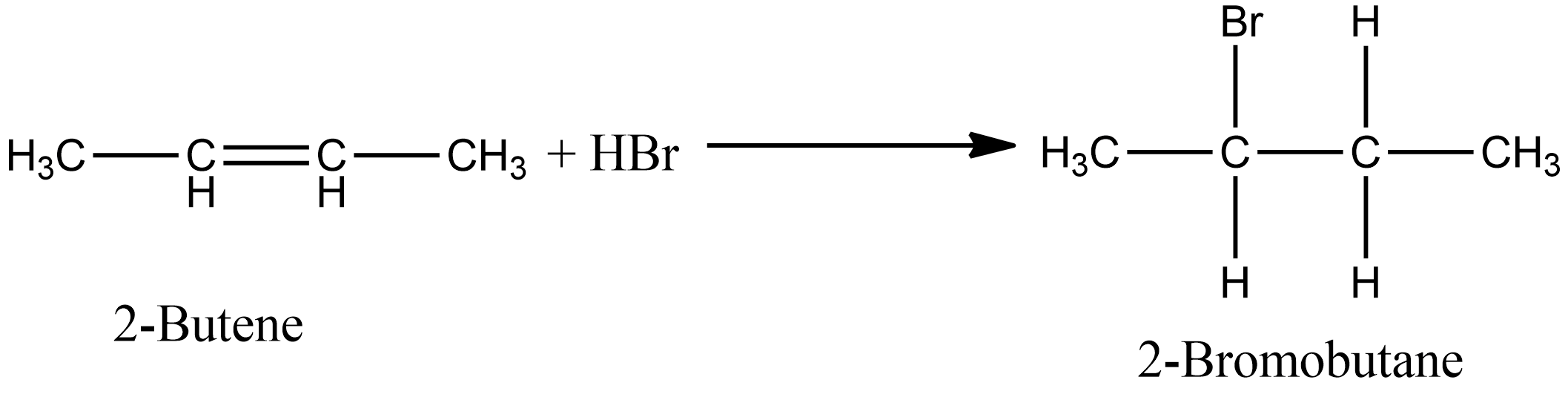
ReasonAddition of HBr on 2-butene follows Markovnikov's rule.AssertionAddition of HBr on 2-butene gives two isomeric products.

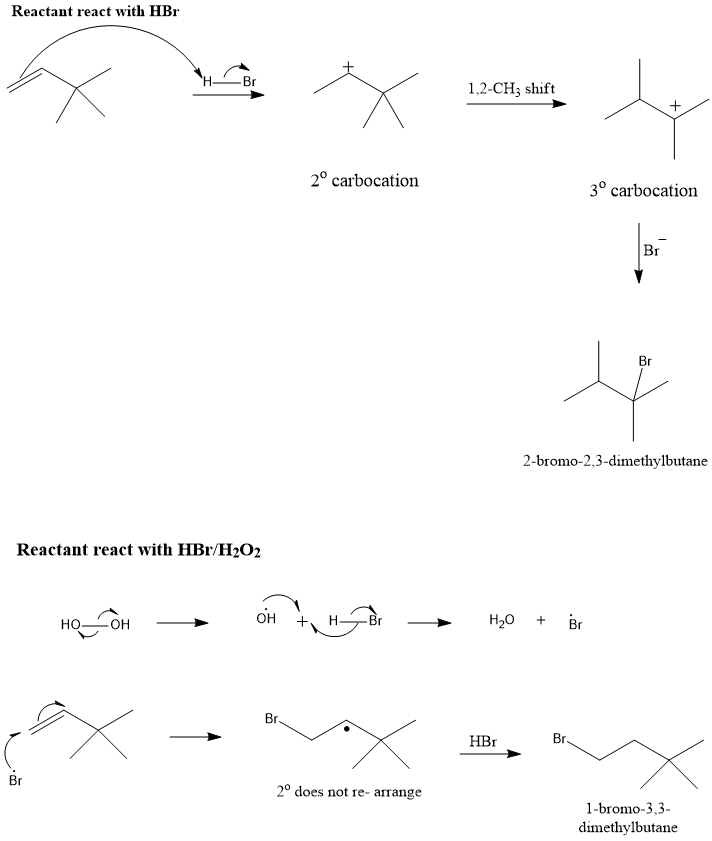
Consider the addition of HBr to 3,3-dimethyl-1-butene, what is the mechanistic explanation the formation of observed product?
Which one of the following compounds will give in the presence of peroxide a product different from that obtained in the absence of peroxide.A. 1- butene,$HCl$B. 1-butene, $HBr$C. 2-butene, $HCl$D. 2-butene, $HBr$
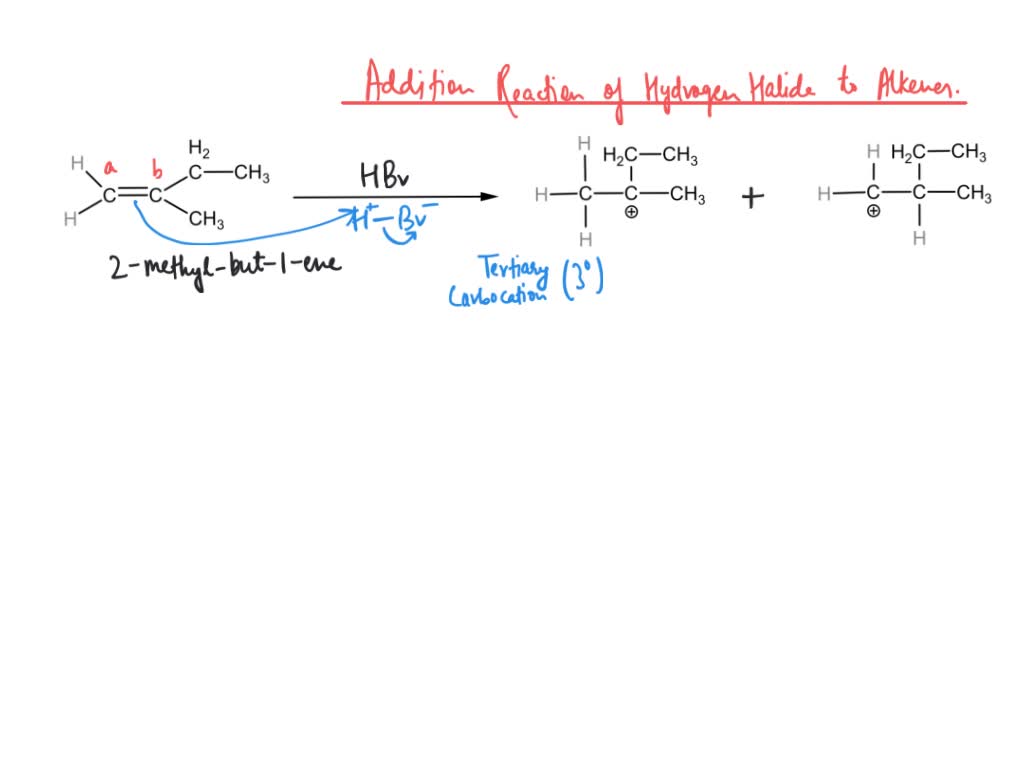
SOLVED: In the reaction of 2-methylbut-1-ene with HBr, there are two possible carbocation intermediates; one is classified as tertiary (3°) and the other as primary (1°). The structures of the starting alkene
A:Addition of HBr in buta 1,3 diene gives 3 bromo but 1 ene as major product at low temperature. R::Addition of HBr in buta 1,3 diene gives 1 bromo but 2 ene
When but 1 en 3 yne reacts with HBr, the H+ adds to the triple bond. Why? If the H+ would have added to the double bond it would have given a

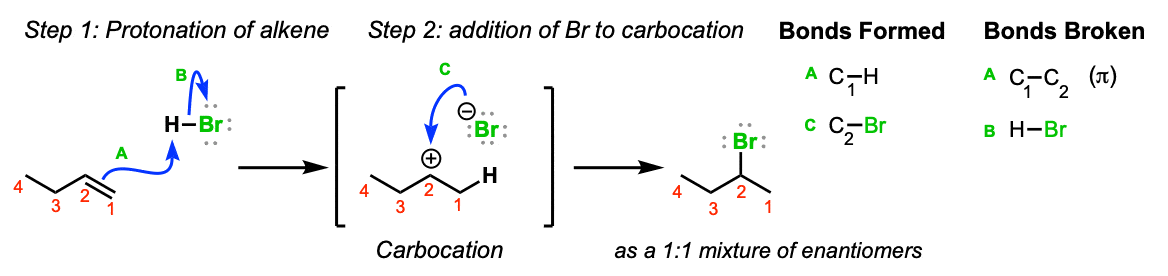
The reaction of 1-butene with HBr in the absence of peroxides yields 2-bromobutane. The mechanism for the reaction involves: A. Isomerization of the 2-bromobutane produced initially B. Attack on the alkene by